North America Active Medical Implantable Devices Market Research: Trends, Opportunities and Industry Forecast
"Executive Summary North America Active Medical Implantable Devices Market Market Size and Share Across Top Segments
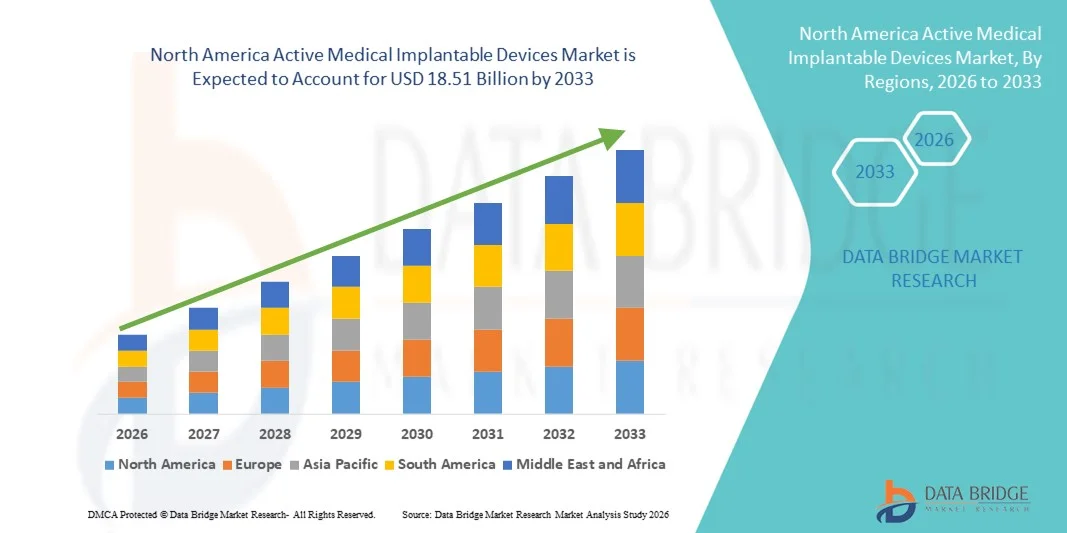
The North America active medical implantable devices market size was valued at USD 10.08 billion in 2025 and is expected to reach USD 18.51 billion by 2033, at a CAGR of 7.9% during the forecast period
This North America Active Medical Implantable Devices Market Market report is composed of a myriad of factors that have an influence on the market and include industry insight and critical success factors (CSFs), market segmentation and value chain analysis, industry dynamics, market drivers, market restraints, key opportunities, technology and application outlook, country-level and regional analysis, competitive landscape, company market share analysis, and key company profiles. Moreover, businesses can gain insights into profit growth and sustainability programs with this report. Business intelligence is an essential aspect when it comes to accomplishing thorough and wide-ranging market insights, and the same is applied for generating this North America Active Medical Implantable Devices Market Market research report.
The North America Active Medical Implantable Devices Market Market report studies various parameters throughout the report, which analyzes the market status in detail. As today’s businesses greatly demand the market research analysis before taking any verdict about the products, opting for such North America Active Medical Implantable Devices Market Market research report is vital for the businesses. Market segmentation gives you a clear idea about the product consumption based on several factors that include, but are not limited to, type, application, deployment model, end user, and geographical region. A reliable North America Active Medical Implantable Devices Market Market research report extends your reach to the success that you desire in your business. So, this global North America Active Medical Implantable Devices Market Market research report is a proven source to gain valuable market insights and take better decisions about the important business strategies.
Review comprehensive data and projections in our North America Active Medical Implantable Devices Market Market report. Download now:
https://www.databridgemarketresearch.com/reports/north-america-active-medical-implantable-devices-market
North America Active Medical Implantable Devices Market Market Growth Snapshot
Segments
- On the basis of product type, the North America active medical implantable devices market can be segmented into implantable cardioverter defibrillators, cardiac resynchronization therapy devices, implantable cardiac pacemakers, neurostimulators, implantable hearing devices, ventricular assist devices, implantable heart monitors, and others. Implantable cardioverter defibrillators are expected to hold a significant share in the market due to the rising prevalence of cardiovascular diseases in the region.
- Based on the material, the market can be categorized into metallic, ceramic, polymeric, and others. The metallic segment is anticipated to dominate the market as these materials offer high durability and strength in medical implants.
- Regarding application, the market can be divided into cardiovascular, neurological, orthopedic, dental, and others. The cardiovascular segment is likely to lead the market owing to the high incidence of heart-related disorders in North America.
Market Players
- Medtronic
- Abbott
- Boston Scientific Corporation
- BIOTRONIK
- LivaNova PLC
- Terumo Corporation
- Med-El
- Cochlear Ltd.
- Sonova
- William Demant Holding A/S
The North America active medical implantable devices market is witnessing significant growth due to the increasing prevalence of chronic diseases in the region, technological advancements in the field of medical implants, and the rising geriatric population. Moreover, favorable reimbursement policies for implant procedures and the presence of a well-established healthcare infrastructure are further driving the market growth. The implantable cardioverter defibrillators segment is expected to witness substantial growth due to the rising cases of arrhythmias and sudden cardiac arrests in North America.
Factors such as the growing adoption of minimally invasive surgical procedures, advancements in implant materials and technologies, and increasing healthcare expenditure are propelling the market forward. Additionally, the rise in awareness about the benefits of active medical implantable devices among patients and healthcare professionals is boosting market demand. However, stringent regulatory approvals and the high cost of implantable devices may hinder market growth to some extent.
The key players in the North America active medical implantable devices market are focusing on strategic initiatives such as product launches, partnerships, acquisitions, and collaborations to strengthen their market positions. For instance, Medtronic recently launched a next-generation cardiac resynchronization therapy defibrillator to cater to the increasing demand for advanced cardiac care products. Collaborations between key market players and research institutions for developing innovative implantable devices are also expected to drive market growth.
The North America active medical implantable devices market is poised for continued growth driven by several key factors. One noteworthy trend is the increasing focus on personalized medicine and patient-specific treatment approaches. As advancements in technology enable greater customization of medical implants to suit individual patient needs, there is a growing demand for active medical implantable devices that can deliver targeted therapies with enhanced efficacy and precision. This shift towards personalized medicine is expected to drive innovation in the development of next-generation implantable devices that offer improved outcomes and patient satisfaction.
Another significant factor shaping the market landscape is the rising trend of remote monitoring and telemedicine in healthcare. Active medical implantable devices equipped with remote monitoring capabilities allow healthcare providers to track patient health metrics in real-time, enabling early intervention and proactive management of chronic conditions. The integration of telemedicine platforms with implantable devices enhances patient monitoring, facilitates timely communication between patients and healthcare professionals, and ultimately improves patient outcomes. This convergence of medical implants with digital health technologies is creating new opportunities for market players to offer seamless and connected healthcare solutions.
Furthermore, increasing investments in research and development activities are driving advancements in implant materials, design, and functionality. Market players are investing in the development of biocompatible materials, such as bioresorbable polymers and smart coatings, to enhance the biocompatibility and longevity of implantable devices. Additionally, the integration of sensor technologies, artificial intelligence, and data analytics in medical implants is enabling the collection of real-time physiological data and the delivery of personalized treatment algorithms. These technological innovations are revolutionizing the field of active medical implantable devices and paving the way for a new era of precision medicine.
Moreover, the evolving regulatory landscape and standards for medical devices are influencing market dynamics and shaping industry practices. Market players are increasingly focusing on ensuring compliance with stringent regulatory requirements and quality standards to mitigate risks and maintain consumer trust. Collaborations between industry stakeholders, regulatory bodies, and healthcare institutions are essential to drive innovation, address regulatory challenges, and ensure the safe and effective deployment of active medical implantable devices in clinical settings. By fostering a collaborative ecosystem focused on patient safety and product quality, market players can navigate regulatory complexities and accelerate the development and commercialization of innovative implantable technologies for improved patient care.
In conclusion, the North America active medical implantable devices market is undergoing rapid transformation driven by technological advancements, shifting healthcare paradigms, and evolving consumer preferences. Market players that embrace innovation, foster strategic partnerships, and prioritize regulatory compliance are well-positioned to capitalize on the growing demand for personalized, connected, and efficacious implantable solutions. As the market continues to evolve, collaboration, adaptation, and a patient-centric approach will be key to driving sustainable growth and delivering value to patients, healthcare providers, and stakeholders across the healthcare ecosystem.The North America active medical implantable devices market is a highly dynamic and competitive landscape characterized by significant growth opportunities driven by various key factors. One notable trend shaping the market is the increasing focus on personalized medicine and patient-specific treatment approaches. Advances in technology have paved the way for greater customization of medical implants to cater to individual patient needs, leading to a surge in demand for active implantable devices that can deliver targeted therapies with enhanced efficacy and precision. This trend towards personalized medicine is driving innovation in the development of next-generation implantable devices that promise improved outcomes and increased patient satisfaction.
Another crucial factor influencing the market is the growing adoption of remote monitoring and telemedicine in healthcare. Active medical implantable devices integrated with remote monitoring capabilities enable healthcare providers to monitor patient health parameters in real-time, facilitating early intervention and proactive management of chronic conditions. The convergence of telemedicine platforms with implantable devices is enhancing patient monitoring, facilitating seamless communication between patients and healthcare professionals, and ultimately improving patient outcomes. This integration of medical implants with digital health technologies presents new opportunities for market players to offer integrated and connected healthcare solutions that enhance patient care and outcomes.
Additionally, investments in research and development are driving advancements in implant materials, design, and functionality. Market players are focusing on developing biocompatible materials and incorporating sensor technologies, artificial intelligence, and data analytics in implantable devices to collect real-time physiological data and deliver personalized treatment algorithms. These technological innovations are revolutionizing the field of active medical implantable devices, ushering in a new era of precision medicine where therapies are tailored to individual patient needs and optimized for superior outcomes.
Furthermore, the evolving regulatory landscape and standards for medical devices are shaping industry practices and driving compliance with stringent requirements. Market players are prioritizing regulatory compliance to mitigate risks, ensure product quality, and maintain consumer trust. Collaborations between industry stakeholders, regulatory bodies, and healthcare institutions are essential to navigate regulatory challenges, foster innovation, and ensure the safe and effective deployment of active medical implantable devices in clinical settings. By fostering a collaborative ecosystem focused on patient safety and product quality, market players can accelerate the development and commercialization of innovative implantable technologies that cater to the evolving needs of healthcare consumers.
In conclusion, the North America active medical implantable devices market is poised for continued growth fueled by technological advancements, the shift towards personalized medicine, and the integration of digital health technologies. Market players that embrace innovation, strategic partnerships, and regulatory compliance are well-positioned to capitalize on the expanding demand for personalized and connected implantable solutions. By staying attuned to market trends, fostering collaboration, and prioritizing patient-centric approaches, stakeholders in the active medical implantable devices market can drive sustainable growth, deliver value to patients and healthcare providers, and contribute to advancements in healthcare delivery and patient outcomes.
Get a closer look at the company’s market penetration
https://www.databridgemarketresearch.com/reports/north-america-active-medical-implantable-devices-market/companies
Global North America Active Medical Implantable Devices Market Market – Segmentation & Forecast Question Templates
- What is the market size snapshot for the North America Active Medical Implantable Devices Market industry?
- What is the global market growth trend for North America Active Medical Implantable Devices Markets?
- Which key segmentations are assessed in the North America Active Medical Implantable Devices Market Market?
- What are the names of top-rated players in the North America Active Medical Implantable Devices Market Market sector?
- What countries offer the highest opportunities in North America Active Medical Implantable Devices Market Market?
- What are the names of leading regional competitors in North America Active Medical Implantable Devices Market Market?
Browse More Reports:
Asia-Pacific Heart Pump Devices Market
Middle East and Africa Heart Pump Devices Market
North America ChloroHydroxyPropylTrimethyAmmonium Chloride (CHPTAC) Market
Europe ChloroHydroxyPropylTrimethyAmmonium Chloride (CHPTAC) Market
Asia-Pacific ChloroHydroxyPropylTrimethyAmmonium chloride (CHPTAC) Market
Middle East and Africa ChloroHydroxyPropylTrimethyAmmonium Chloride (CHPTAC) Market
North America Distributed Antenna System (DAS) Market
North America Road Safety Market
Europe Road Safety Market
Asia Pacific Smart Lighting Market
Middle East and Africa Smart Lighting Market
Asia-Pacific Silicone Textile Chemicals Market
Europe Silicone Textile Chemicals Market
Middle East and Africa Silicone Textile Chemicals Market
North America Silicone Textile Chemicals Market
About Data Bridge Market Research:
An absolute way to forecast what the future holds is to comprehend the trend today!
Data Bridge Market Research set forth itself as an unconventional and neoteric market research and consulting firm with an unparalleled level of resilience and integrated approaches. We are determined to unearth the best market opportunities and foster efficient information for your business to thrive in the market. Data Bridge endeavors to provide appropriate solutions to the complex business challenges and initiates an effortless decision-making process. Data Bridge is an aftermath of sheer wisdom and experience which was formulated and framed in the year 2015 in Pune.
Contact Us:
Data Bridge Market Research
US: +1 614 591 3140
UK: +44 845 154 9652
APAC : +653 1251 975
Email:- corporatesales@databridgemarketresearch.com
"
- North_America_Active_Medical_Implantable_Devices_Market
- North_America_Active_Medical_Implantable_Devices_Market_size
- North_America_Active_Medical_Implantable_Devices_Market_share
- North_America_Active_Medical_Implantable_Devices_Market_forecast
- North_America_Active_Medical_Implantable_Devices_Market_trends
- North_America_Active_Medical_Implantable_Devices_Market_companies
- Art
- Causes
- Crafts
- Dance
- Drinks
- Film
- Fitness
- Food
- Jocuri
- Gardening
- Health
- Home
- Literature
- Music
- Networking
- Alte
- Party
- Religion
- Shopping
- Sports
- Theater
- Wellness


